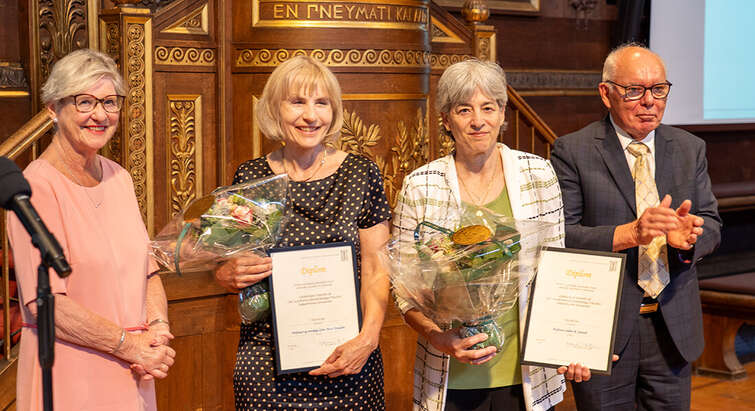Professor Miriam Kolko, Department of Drug Design and Pharmacology, and Postdoc Catharina Thiel Sandholdt, Department of Public Health, have recently received grants from VELUX FOUNDATION within the area ”transitions and changes during the adult life”.